What is the Aufbau Principle?
The Aufbau Principle was formulated during the 1920s by Wolfgang Paul and Neil Bohrs. If we understand the semantics of the word “Aufbau” it means “building up” in German. The principle deals with various laws around the filling up of electronic orbitals.
Atomic orbitals are of different types. Some orbitals are higher energy in nature and some are lower energy in nature. The Aufbau principle states that in a neutral state, the electrons are filled in a specific order wherein electrons always occupy the lowest energy levels.
After all the available lower energy orbitals are filled the higher energy orbitals are filled. This helps us in providing a pattern around the process through which electrons are filled into atomic orbitals.
Atomic orbitals
The Heisenberg Uncertainty Principle states that it’s improbable to understand an electron and determine its future course of action. However subsequent research around this domain has helped us understand the probable region where electrons exist. Thus we use atomic orbitals to understand the possible position of an electron around the nucleus m
We already know that electrons revolve around the nucleus. However, these electrons exist in pairs and are located at various distances from the nucleus. Atomic orbitals help us gain insights into this phenomenon. The atomic orbitals of any atom are unique to themselves.
They are considered to be the vast physical space around the nucleus. The mathematical functions associated with an atomic orbital help us understand the probable location where the electron might be present.
Types of orbitals
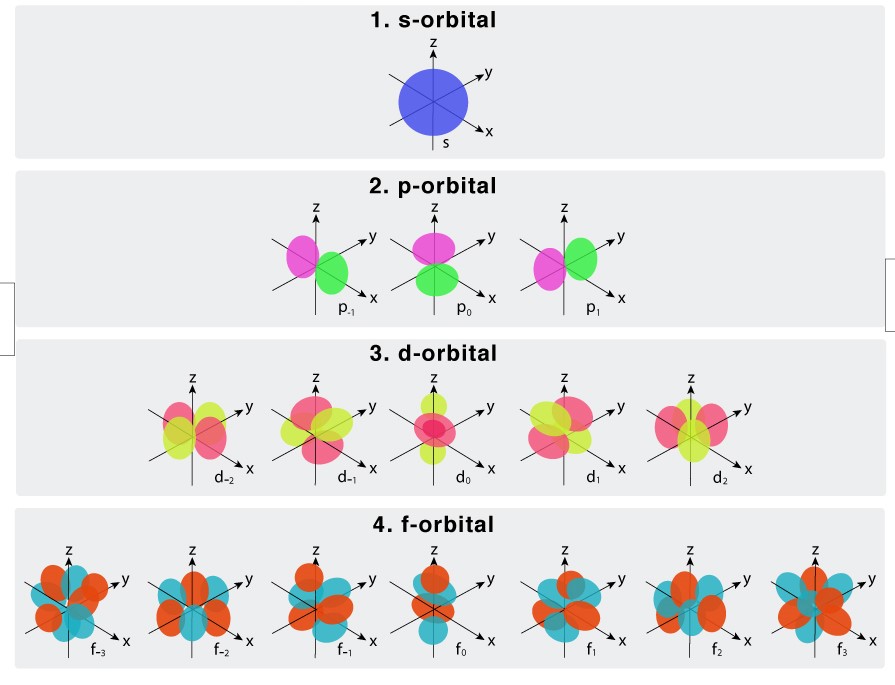
Atomic orbitals are classified into four types. Each type has its characteristics that influence the atom in general. The four types of orbitals are s/p/d/f. Each orbital has a specific capacity for holding electrons. While the ‘s’ orbital holds two electrons other orbitals can hold a higher number of electrons.
Capacity of orbitals
The ‘S’ orbital can occupy two electrons. The ‘P’ orbital possesses the capacity to occupy six electrons. The ‘D’ orbital can accommodate a total of ten electrons. The ‘F’ orbital can occupy a total of fourteen electrons.
Electronic configuration
The electronic configuration of an atom is dependent on the distribution of various electrons into the atomic orbitals. An atom’s electronic configuration helps us understand the reactivity and interaction between elements. The goal of any element is to attain a stable electronic configuration.
A stable electronic configuration is a situation wherein each atomic orbital is filled with the respective number of electrons. Therefore the reason for any reactivity within an element is due to unstable electronic configuration. The instability propels an atom to make or break bonds with other atoms.
Understanding Orbitals
Electrons occupy orbitals around an atom. Each specific orbital possesses a specific energy level. Orbitals are of four types. Let’s understand these orbitals and their types. The orbitals determine the physical proximity to the nucleus of an atom.
These orbitals are typically classified into s/p/d and f. However, each of these four levels is further divided into sub-levels.
Aufbau Principle
The Aufbau Principle simply states that electrons start filling themselves starting from the lower energy levels and move forward to higher energy levels. This helps generate a systematic approach towards the filling of electrons wherein the filling process is directly dependent on the energy levels. The electrons start filling the lower energy levels followed by their sublevels and thus keep progressing ultimately. The reason behind filling electrons based on relative energy can be attributed to the basic nature of an electron which focuses on always generating stability.
The Aufbau principle provides us with the sequence through which electrons start filling around the orbitals. The sequence is as follows:
1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s……
Understanding the sequence
The energy level ‘n=1’ is associated with the 1s orbital. Thus it always accommodates a total of 2 electrons. The energy level ‘n=2’ is associated with 2s and 2p electrons. The ‘s’ orbital has a capacity of two electrons while the ‘p’ orbital has a capacity of six electrons.
Thus the level ‘n=2’ accommodates a total of eight electrons. The energy level ‘n=3’ is associated with a single ‘s’ orbital, three ‘p’ orbitals, and five ‘d’ orbitals.
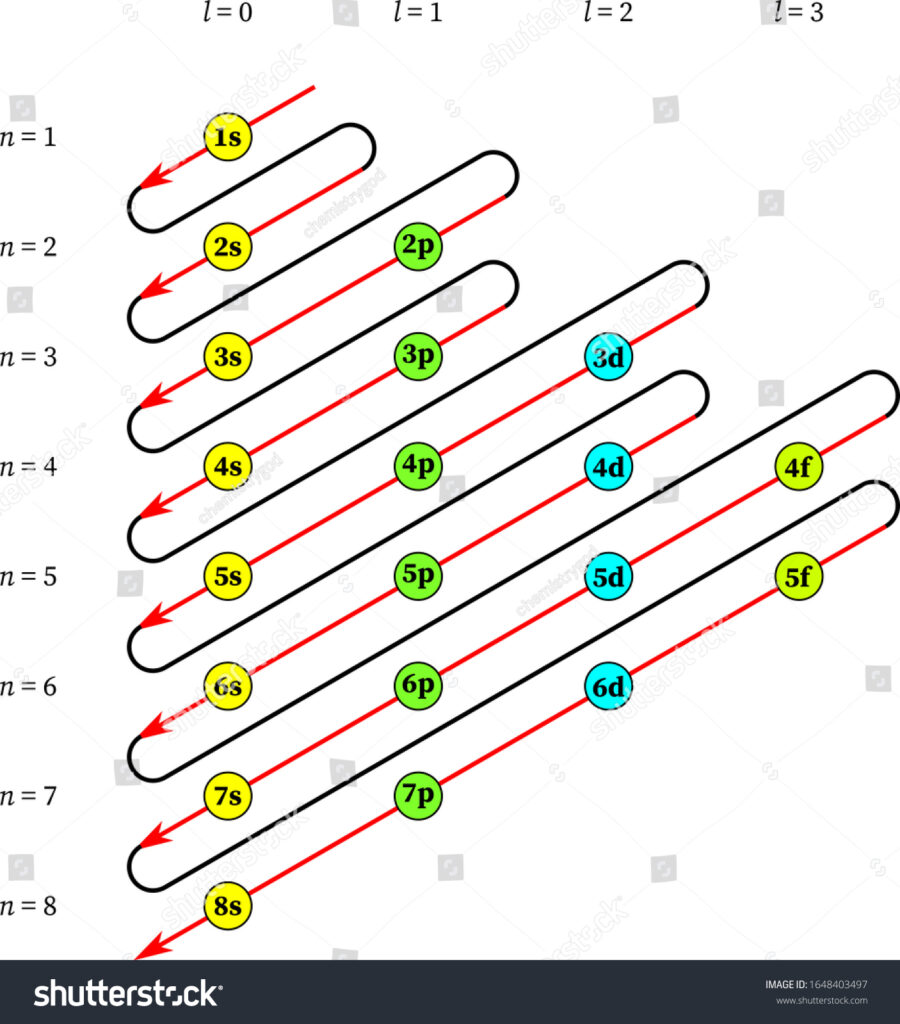
This sequence dictates the filling order which can be understood according to the image sequence given above. The Aufbau principle directs this filling order.
Let’s take an example to understand this in detail. A carbon atom has six electrons. These electrons will be filled in a specific manner. The first two electrons will occupy the ‘1s’ orbital which is closest to the nucleus. The next electrons will immediately occupy the ‘2s’ orbitals. The remaining electrons will try to integrate themselves in the ‘3s’ energy level. The ‘3s’ energy level needs six electrons to stabilize itself but it’s only filled with two electrons. Therefore carbon always tries to form bonds and attain stability either by donating its two electrons or trying to take four electrons from other atoms.
How Material Sciences Used the Aufbau Principle
Material science deals with elements and atoms. The properties of any atom or element directly depend on the electrons which influence the nature and reactivity of any atom. Even the internal arrangements of electrons within any atom is of paramount importance to understanding the characteristics of any atom.
The Aufbau rule directly provides principles that can help predict various features of any atom. Some of its uses are as follows:
Elemental Properties
The electronic configuration of any atom can be determined with the help of the Aufbau Principle. The principle gives us a direct process that can help in predicting the electronic configuration of any atom by taking into account its atomic number. The understanding of the atomic number helps in understanding the chemical nature and reactivity of any atom.
- The Valence electrons of an atom reside in the outermost energy levels of an atom. These electrons help in determining the reactivity of an atom. Any electron with a similar number of valence electrons always displays a similar behavior regarding reactivity or bond formation.
Similarly, various characteristics of an atom such as reactivity, atomic size, or ionization energy can be determined with the help of the Aufbau principle.
Advanced Materials
The characteristics of any atom can be determined with the help of its electronic configuration. The Aufbau principle which helps in determining the electronic configuration has been instrumental in research focused on design elements.
Modern-day research has been focused on crafting specific elements by tailoring various electrons in an atom. This process helps scientists design materials with specific characteristics based on their ease. These materials are designed with exceptional fine-tuning around various aspects such as conductivity, magnetism, thermal stability, and various optical properties. Some examples of such materials are as follows:
- Superconductors
A superconductor is a spectacular material that has zero resistance to electricity at lower temperatures. This has resulted in the heavy use of superconductors for various applications such as power transmission or the development of advanced electrical devices.
A phenomenon known as ‘Cooper pairing’ is referenced to electrons forming pairs and moving through any material without experiencing scattering. Various materials such as Yttrium-Barium-Copper Oxide (YBCO) or Niobium-Titanium (Nb-Ti) have been specifically engineered by manipulation of their electronic configurations. By optimizing their electronic configuration these materials have been made conducive for Cooper pairing.
- Photovoltaic material
Solar cell technology has a heavy use of photovoltaic material. The material is specifically used to convert light into solar energy. With the help of extensive research around elements and by controlling their electronic configuration various materials have been designed wherein sunlight is absorbed efficiently and the flow of electrons is generated seamlessly.
Cadmium Telluride (CdTe) and Copper Indium Gallium Selenide (CIGS) are materials that have been severely optimized to initiate maximum optimization of the bandgaps of the electromobility. This helps in heavily increasing the efficiency of the solar cell.
Thus we can understand that the entire research around modern material science is based on elements that direct us back to atoms. Electrons play a major role in influencing the characteristics and properties of any atom. The Aufbau principle deals with electrons and provides specific guides which aid in understanding electrons. Thus we can understand how the Aufbau principle forms the defining principle around which material scientists research and understand materials and elements while trying to understand their various properties.
Nanotechnology and Aufbau Principle
Nanotechnology has recently developed itself into a defining branch of science that has heavily focused on designing and production of various devices or structures by tweaking the structure of atoms or molecules at a nanoscale level. Nanotechnology has bought a revolution in the way we see matter. The intentional manipulation of atoms or molecules at a nanoscale level has enabled the design and development of extraordinary materials with unique properties.

The Aufbau principle provides insights into electronic configuration which serves as a beacon for nanotechnology research which is heavily dependent on electronic configurations. The insights provided into the behavior of electrons helps provide researchers with a blueprint of the atom before undertaking the tweaking process. The uses of nanotechnology are as follows.
Manipulation of electronic properties
The behavior of the atom at the nanoscale is directly influenced by the electrons and their arrangement process. The electrons of an atom can be specifically engineered to provide desired effects by using the Aufbau principle.
- Quantum dots
An example of such a technological marvel is the ‘Quantum dots ie. A nanoscale semiconductor. This semiconductor specifically controls the arrangement of the electrons within these quantum dots and produces definite results. The process of confining the electrons in a restricted space and fine-tuning them as per your requirements helps in the manipulation of electronic and optical properties.
This helps in crafting nanomaterials with a custom emission and absorption process. These semiconductors are used in the domain of optoelectronics and quantum computing.
- Nanowires and Nanotubes
A one-dimensional structure that possesses extraordinary properties is known as nanotubes or nanostructures. The arrangement of the electrons inside these structures is done with the help of the Aufbau principle.
Manipulation of the electronic properties of these nanowires or nanotubes helps in engineering various such structures with enhanced conductivity and various characteristics. These engineering processes have accelerated the development of various sensors or nanorobots which are high-performance in nature
- Quantum effects
The behavior of any material is guided by quantum effects at a nanoscale level. This directly affects the behavior of any material which results in the formation of unique properties. The beacon light in the research around quantum effects is the Aufbau principle.
Manipulation of any electronic configuration results in the creation of various nanostructures.
These tailored structures result in the creation of various effects such as quantum coherence and quantum confinement. These effects further help in the development of advanced technology and science focused around quantum computing and quantum communication which aid in developing secure communication and transmission lines.
The Aufbau principle directly influences electron behavior however there are various limitations while applying these concepts in nanotechnology.
- Quantum mechanics
The Aufbau principle provides a theoretical framework for the electronic configuration of various elements. Various predictions about the nature of elements can be conducted with the help of the Aufbau principle. However quantum mechanics introduces various limitations in itself which results in the predictions turning into a bluff. The quantum mechanics models are limited in itself and as a result, the accuracy of predictions through the Aufbau principle is severely impacted.
- Experimental accuracy
The Aufbau principle provides various predictions about electronic configuration based on a theoretical framework. Although they might be valid through a natural science perspective it’s completely different when we view it through a nanotechnology-based approach. Nanotechnology is heavily research based and it thrives on experimental validation.
Various assumptions by the Aufbau principle need to be verified by experiments which will ensure their real-world applicability. While conducting such testing there are instances wherein the assumption turns out to be a misappropriation due to various factors in play.
- Material specific deviations
The Aufbau Principle provides simple models based solely on theory but it doesn’t take material specific consideration in the big picture. Various considerations such as defects, doping, and various quantum confinement-specific effects need to be taken into consideration. Blatantly ignoring these material-specific considerations might affect the overall engineering of materials from a nanoscale perspective.
A closer look at material science and nanotechnology reveals how the Aufbau principle has been a guiding principle in helping modern scientists unravel the secrets of elements and atoms. Not only has the principle been instrumental in the past but it has served as a guiding light for nanotechnology in its quest for perfection through a nanoscale perspective.
Although it has its share of limitations it’s still completely relevant and serves as a guiding beacon light for future research work and acquaints scientists with the very basics of elements and atoms and the interplay between electrons and chemical reactions.
